Definitions and FAQ
inTERP FAQ
Faculty and Exempt Staff - reporting Outside Professional Activities
When is the Go-Live date?
The go-live date is January 12th, 2026.
Is this a new requirement?
No, the University of Maryland Policy on Conflict of Interest and Conflict of Commitment requires all faculty and exempt staff to disclose at least annually.
The inTERP system combines the Outside Professional Activities (OPA) reporting system, the previous COI Disclosure System (KCOI), the Office of Faculty Affairs Consulting MOU system for reporting external relationships.
When do I have to disclose?
- Within 30 days of being hired at the University, even if there are NO activities to disclose
- All Faculty, Exempt-Staff, and Graduate Students are required to disclose at least annually AND prior to engaging in a new Outside Professional Activity.
- Updates are required on a rolling basis within 30 days of acquiring (or becoming aware of) a new significant financial interest or beginning a new outside activity
- If you don’t have a disclosure on file, disclose NOW.
How is inTERP reporting different from the Outside Professional Activities System?
inTERP is open year-round, rather than only during a specific period during the year. Updates can be made at any point throughout the year, and each individual’s annual renewal date will be unique.
Questions regarding how Article 25, Section 6 of the 2024 AFSCME and USM MOU applies to AFSCME bargaining unit employees?
Please contact Keisha Williams, Program Director for Labor Relations at kwilli40@umd.edu.
Performing union duties and responsibilities, including serving as an AFSCME job steward, as outlined in the 2024 USM and AFSCME MOU, is not an outside professional activity that must be reported.
Article 5, Section 4 of the USM and AFSCME MOU
Note: This section explains performing union duties including serving as a job steward.
"The Employer and the Union recognize that job stewards play an important role in effectuating the terms of this MOU; however, both parties acknowledge that the
duties undertaken as a job steward are in addition to their job assignments...
Each job steward will be a working employee, who, consistent with the operational needs of their employer, shall be permitted a reasonable amount of time during regular working hours at their campus to:
1. Investigate and Process grievances,
2. Attend and participate in discipline meetings called by the Employer,
3. Attend new employee orientation as permitted by law,
4. Participate in LMC meetings, committee meetings and activities if such meetings or activities have been jointly established by the parties
5. Attend meetings called or agreed to by the Employer if such employees are entitled and required to attend the meetings by virtue of being job stewards,
6. Participate in activities related to the administration of this MOU including problem solving to avoid the filing of grievances, and
7. Attend negotiating sessions regarding supplementation or amendment of this MOU or a local agreement during its term."
Where do I go for resources and questions?
The Disclosure Office website has a wealth of information, and the Disclosure Office Help Center (VPN required) has additional resources for support completing the disclosure. All inTERP questions should be directed to interp@umd.edu.
How do I access inTERP?
Please go to this link to gain access to the inTERP system.
Is there a user guide for inTERP?
The Disclosure Office has developed the following inTERP Guide: For Users
When do I need to disclose?
- New to UMD: Within 30 days of being hired at the University, even if there are NO activities to disclose
- Prior to engaging in a new Outside Professional Activity
- When Submitting a Proposal: As of October 1, 2025, UMD requires a disclosure at the time of submission to propose to ANY entity. Disclosure is required even if there are NO activities to disclose. The disclosure is required, not the review and disposition. The Disclosure must be cleared—reviewed, dispositioned, and managed as needed before AWARD monies can be released.
- Note: A new disclosure is not required for every proposal; however, there must be an up-todate inTERP disclosure on file, i.e., inTERP disclosure less than a year old and/or updated if there have been changes. Researchers should review their disclosure and update as needed.
- Example: A researcher disclosed in July 2024 for an NSF proposal submission. In January 2025, they submitted a new proposal to NIH. A new disclosure is only required if there are changes in their outside activities between July and January. If there have been no changes, the July 2024 disclosure complies with the requirement to have an up-to-date disclosure on file.
- Within 30 Days: Updates required on a rolling basis within 30 days of acquiring (or becoming aware of) a new significant financial interest or beginning a new outside activity
- Be transparent in the disclosure: More details = fewer iterations.
What agencies require disclosure at the time of submission?
Effective October 1, 2025: Disclose to Propose
All individuals named on a proposal—faculty, staff, and graduate students—must have a current disclosure on file in inTERP at the time the proposal is submitted, regardless of the funding source or sponsoring entity.
Note: A new disclosure is not required for every proposal; however, there must be an up-to-date inTERP disclosure on file, i.e., inTERP disclosure less than a year old and/or updated if there have been changes. Researchers should review their disclosure and update as needed.
Example: A researcher disclosed in July 2024 for an NSF proposal submission. In January 2025, they submitted a new proposal to NIH. A new disclosure is only required if there are changes in their outside activities between July 2024 and January 2025. If there have been no changes, the July 2024 disclosure complies with the requirement to have an up-to-date disclosure on file.
The disclosure must be cleared—reviewed, dispositioned, and managed (as needed)—before the award funds are released.
Where can UMD Department and College Administrators confirm compliance for their project personnel?
Department and College Administrators can review compliance for their project personnel via the Disclose to Propose report. (Must be connected to the VPN to access the report).
If a faculty member attempts to submit a proposal, and they have not completed an inTERP disclosure, will ORA not submit it to the sponsor? Will ORA be checking on that?
- ORA checks for disclosure now for all agencies requiring disclosure at the time of submission and/or award.
- If there is NO DISCLOSURE ON FILE, one must be submitted inTERP before the proposal can be submitted.
- Department and College Administrators should use the Disclose to Propose report well in advance of the deadline to ensure compliance.
How do I edit, delete, or create a new version of an inTERP disclosure?
The Disclosure Office has developed a few guides to assist with editing, deleting and creating a new version of disclosures.
How do I disclose a MIPS, MII, MEII, SBIR, or STTR Proposal or Active Award?
Instructions for disclosing a proposal can be found here.
Definitions
What is a Conflict of Interest?
The University of Maryland’s Policy Definition:
The term “conflict of interest” denotes situations in which members of the University community are in a position to gain, or appear to gain, financial advantage or personal benefit (broadly construed) arising from their University positions, either through outside professional activities or through their research, administrative, or educational actions or decisions at the University. Sometimes these conflicts of interest can be managed, and sometimes they cannot be managed and must be avoided; regardless, conflicts of interest must always be recognized, disclosed, and appropriately addressed.
Additional information regarding Conflicts of Interest
Refers to a situation in which financial, professional, or other personal considerations may directly or indirectly affect, or have the potential to affect, an employee’s professional judgment in exercising any university duty or responsibility in administration, management, instruction, research, or other professional activities. This can include interests that bias the nature or direction of scholarly research or influence decisions concerning teaching and students (mentoring), appointments and promotions, use of university resources, interactions with human subjects, or other matters of interest to the university.
Conflicts of Interest also refer to situations where a Significant Interest, including a Significant Financial Interest, could directly and significantly affect the design, conduct, or reporting of research or educational activities.
A Conflict of Interest includes any Financial Conflicts of Interest.
What is the Conflict of Interest Committee?
The advisory committee, appointed by the President, in accordance with the University’s COI/COC Policy and Procedures related to research endeavors.
The COI Committee typically meets on the 2nd Wednesday of the month. The disclosure must be completed, electronically routed, and approved in inTERP by the first of the month for submission to that month's agenda.
What is a Conflict of Commitment?
A circumstance that arises when otherwise acceptable outside research-related activities may compromise or have the potential to compromise the fulfillment of a University employee’s institutional responsibilities, as stipulated under Maryland State Ethics Law and/or otherwise determined by the unit head.
What is Consulting?
Any research-related additional activity beyond a University employee’s institutional responsibilities, professional in nature, and based on the appropriate discipline or area of expertise, for which the employee may receive additional personal remuneration, and where such activities primarily benefit the employee and not the University.
What is a Significant Interest?
A Financial Interest or other personal interest received from or held in an entity outside of the University that reasonably appears to be related to your Institutional Responsibilities. Significant Interest includes Significant Financial Interest.
What is a Financial Conflict of Interest (FCOI)?
A significant financial interest that could directly affect the design, conduct, or reporting of funded research. FCOI includes (but is not limited to) ownership, equity, etc.
What is a Financial Interest?
Anything of value, whether or not the value is readily ascertainable.
What is a significant Financial Conflict of Interest (FCOI)?
Typically defined as the value of any remuneration (salary, payment for services, consulting fees, honorarium, paid authorship, equity, option). The UMD threshold for reporting a FCOI is $0 as prescribed by Maryland State Ethics Law and University policy.
What is a "Fiduciary Role(s)"?
A role that imparts an obligation (i.e., a fiduciary duty) upon an individual to act on behalf of another person(s) or entity and put those interests, typically financial, ahead of one’s own. Titles that include terms such as executive, officer, director, or manager and positions with titles such as CEO, Director, Scientific Officer, or Vice President are examples of designations that may indicate a role with fiduciary responsibilities.
What is the difference between an actual and potential or perceived conflicts of interest?
An actual Conflict of Interest arises where financial or other personal or professional considerations compromise an individual’s objectivity, professional judgment, professional integrity, and/or ability to perform his or her responsibilities to the University. Perceived or potential Conflicts of Interest exist in situations where an individual member of the University community, a member of the individual’s family, or a close personal relation has financial interests, personal relationships, or professional associations with an outside individual or organization, such that his or her activities within the University could appear to be biased or unduly influenced by that interest or relationship.
What does "reimbursed or sponsored travel" include?
PHS regulations require one to disclose travel paid by:
External professional organizations and societies; academic journals and publishing companies; and ANY travel reimbursed or sponsored by a foreign Institution of higher education or the government of another country.
What does "Institutional responsibilities" mean?
It means an Investigator's professional responsibilities on behalf of the Institution, and as defined by the Institution in its policy on financial conflicts of interest, which may include, for example: activities such as research, research consultation, teaching, professional practice, institutional committee memberships, and service on panels such as Institutional Review Boards or Data and Safety Monitoring Boards.
What does it mean when I have a Dual Relationship?
This occurs when a UMD employee engages in two or more activities with the same entity. For example, an employee is the PI on an active sponsored research agreement with Company A. Company A also wants to provide a gift to support a graduate student in the PI’s lab. This creates a dual relationship between the PI and Company A.
What is a Management Plan?
A document developed with the Disclosure Office and approved by the University President to (1) obtain information and commitments regarding Significant Interests and prevent such Significant Interests from developing into Conflicts of Interest and/or (2) Manage Conflict(s) of Interests for a particular Reporter (Discloser).
The Management Plan may address the areas of university responsibilities implicated, including:
- Objectivity and scientific integrity, including disclosure of a Reporter (Discloser)’s conflict in publications and presentations;
- Objective treatment of students and/or subordinates;
- Objective treatment of, and scientific integrity concerning, human subjects;
- Decisions regarding the development, disclosure, and use of university Intellectual Property;
- Decisions regarding the decision to purchase products or services; and
- Decisions regarding the use of the Reporter (Discloser)’s time as well as other resources of the university
What are the four basic interests related to conflicts?
You can categorize an individual's potential interests into these four types:
- Direct interests: These include an individual's own personal self-interest, family obligations, and personal business interests.
- Indirect interests: These include the personal, family, and business interests of people or groups with whom the individual associates.
- Financial interests: These include anything of monetary value, including services rendered, commercial dealings, or equity ownership.
- Non Financial interests: These involve personal or familial relationships and other potential sources of bias.
What are the five common types of Conflicts of Interest?
1.) Self-Dealing: Self-dealing occurs when an official acts in their own best interest in a transaction at the expense of the organization or their clients. They may use the organization's money or access for personal betterment at the expense of the business. It represents an illegal act that can lead to litigation, penalties, and termination of employment for those who commit self-dealing.
2.) Familial relationships (Nepotism): Familial relationships may result in favoritism for family members or friends. In this situation, a person with power at an organization awards favors, benefits or opportunities to their family members based on the relationship rather than actual qualifications.
3.) Excess Compensation: Most common in the nonprofit sector, excess compensation occurs when the organization pays an employee far more than a person in a comparable role in another organization. The employee is usually high-ranking, with substantial social or political influence.
4.) External Employment: When someone works more than one job in the same sector, a conflict of interest can arise. If working for one company provides you with access to proprietary information. If you use that information for your second job, that's a conflict of interest. This is especially true if you've signed a non-disclosure agreement.
5.) Gifts: Accepting gifts from external interested parties to gain an advantage is a conflict of interest. While sometimes people give gifts without an ulterior motive, the acceptance of a gift can improperly influence someone's judgment.
Conflict of Interest Questions
What do I need to report?
You are required to disclose all Significant Interests held by you or a close family member related to or that reasonably appear to be related to your institutional responsibilities. In addition, you must provide information about the sources of funds that support your research.
Examples of Significant Interests you need to disclose include, but are not limited to:
- External affiliations, activities, & appointments
- Positions for which compensation is received
- Corporate Board membership, paid or unpaid
- Ownership interests
- Equity, stock, stock options in a publicly traded entity or in a non-publicly traded entity of any value
- External consulting activities and other outside work
- any service that is 1) related to your professional expertise in your university position, 2) provided to an external entity, 3) for remuneration (monetary) or other compensation from a single entity, 4) including but not limited to advising; conducting research or analysis; writing reports; delivering presentations or training; contributing to or managing projects; designing, developing or producing products or services; providing clinical services
- Foreign activities of any kind. A research collaboration with the intent to publish with individuals at any foreign entity, organization and/or institution from which you received monetary or non-monetary value (including free or subsidized space, laboratories, equipment, or materials; access to otherwise restricted data; support services; visiting researchers or personnel).
- A research collaboration may include, but is not limited to, one or more of the following:
- Participating in the research design
- Collecting, sharing or analyzing data
- Sharing research results prior to pre-publication editorial review
- Designing, fabricating, improving or testing equipment together
- Traveling in support of the above actions
- A research collaboration may include, but is not limited to, one or more of the following:
- In-kind contributions (anything of value not in the form of cash) from an external entity with an associated time commitment. This includes, but is not limited to, free or subsidized use of space/offices/laboratories, equipment, materials, support services, visiting researchers or other personnel, intellectual property rights, sponsored travel, etc.
- Reimbursed or sponsored travel
- To a U.S. destination regardless of value per entity (except travel that is reimbursed or sponsored by the University of Maryland)
- To a foreign destination of any value
- Intellectual property
- Licensing or royalty income
- Foreign talent recruitment program participation
When do I disclose an actual or perceived Conflict of Interest and/or Commitment?
Disclosures must be submitted at least annually and updated within thirty (30) days of a Change in Status of a previously disclosed Significant Interest or discovering or acquiring a new one. Reporters (Disclosers) must include any information not previously disclosed in any other disclosure period as well as updated information regarding any previous disclosures.
How can I avoid a Conflict of Interest?
In order to avoid Conflicts of Interest, you must:
- Disclose your duties in the best interests of the university and in such a manner that professional judgment in administration, management, instruction, research, and other professional activities are not affected by any outside financial and/or personal interests;
- Recognize your University appointment as your primary position; and
- Conduct your affairs so as not to derive private gain from your association with the university, except as provided by policy, including but not limited to:
- Not assisting any person and/or organization for any fee or other compensation in obtaining any contract, license, or other economic benefit from the university,
- Not receiving material compensation from others for performance of university duties, and
- Not using university property and/or resources for personal gain except as authorized by university policy.
Should you encounter any potential activity(ies) for which expectations and responsibilities are unclear or that involve foreign affiliations, email the COI Office at COI@umd.edu and/or your Supervisory Authority to discuss how to address the issue.
Note: Some conflicts cannot be avoided, but must be removed or reduced via a Management Plan that will be individually developed based on the disclosure.
What if I am unsure about whether a particular situation amounts to an actual or perceived Conflict of Interest or Commitment?
The Reporter (Discloser) does not decide whether a conflict exists.
- It is your responsibility to report Significant Interests completely, accurately and in a timely fashion.
- It is the responsibility of the COI Committee to evaluate Disclosures of Significant (Financial) Interests, determine if any Conflicts of Interest exist, and support the development, if applicable, of a Management Plans for Significant Interests and/or Conflicts of Interest.
- While the COI Office is responsible for drafting and collecting information relevant to a Management Plan, final authority for authorizing and approving the Management Plan rests solely with the University President.
If I disclose, will I be prohibited from participating in the activity?
A common misconception is that people think disclosing a potential conflict means there necessarily IS a conflict and that if there is a conflict they are not allowed to do the activity. This is not the case, the COI Committee is responsible for developing management strategies (where possible) to manage potential, perceived or real conflicts.
Are there ramifications if I do not comply?
Conflict of Interest or Commitment non-compliance includes, but is not limited to:
- Failure to disclose Conflicts of Interest and/or Commitment;
- False or inadequate Disclosure of Conflicts of Interest and/or Commitment;
- Failure to adhere to a prescribed Management Plan; and
- Failure to adhere to training or reporting requirements.
If the COI Committee determines that an alleged Conflict of Interest or Commitment violation(s) has occurred, the COI Office will refer the matter to the Supervising Authority for investigation under applicable campus supervisory and/or conduct processes and for potential sanctions. The COI Office will assist with the matter as requested.
If the alleged violation, including failure of a Reporter (Discloser) to comply with the COI/COC Policy or a Conflict of Interest Management Plan, appears to have biased the design, conduct, or reporting of sponsored research, as prescribed by law the university will comply with any notification and reporting requirements regarding such bias to the applicable sponsor. For example, for situations involving PHS-funded research, the university will promptly notify the PHS Awarding Component of the corrective action taken or to be taken. The specifics of potential sanctions depend on University policy and the funding authorities.
I'm being asked if I and/or my family members have an equity interest or have received renumeration in any publicly or non-publicly traded entity. Do investments accounts qualify?
Under the Remuneration and Equity Interest question, the Reporter (Discloser) is not mandated to include the following: Income from investment vehicles, such as mutual funds and retirement accounts, as long as you do not directly control the investment decisions made in these vehicles.
What is the difference between an Individual (Personal) Conflict of Interest (COI), a Financial Conflict of Interest (FCOI), Institutional Conflict of Interest (ICOI), and an Organizational Conflict of Interest (OCI)?
Personal Conflict of Interest (COI) - Personal Conflicts of interest can occur when members of the University community are in a position to gain or appear to gain, financial advantage or personal benefit (broadly construed) arising from their University positions, either through outside professional activities or through their research, administrative, or educational actions or decisions at the University. Because University employees are also State employees, provisions in the Maryland Public Ethics Law and University Policies in regard to outside activities still apply. Federal regulations may also apply when an activity is federally funded.
Refers to a situation in which financial, professional, or other personal considerations may directly or indirectly affect, or have the potential to affect, an employee’s professional judgment in exercising any university duty or responsibility in administration, management, instruction, research, or other professional activities. This can include interests that bias the nature or direction of scholarly research or influence decisions concerning teaching and students (mentoring), appointments and promotions, use of university resources, interactions with human subjects, or other matters of interest to the university.
Conflicts of Interest also refer to situations where a Significant Interest, including a Significant Financial Interest, could directly and significantly affect the design, conduct, or reporting of research or educational activities.
Financial Conflict of Interest (FCOI) - Is a form of personal conflict of interest involving a significant financial interest that could directly affect the design, conduct, or reporting of funded research. FCOI includes (but is not limited to) ownership, equity, etc. Typically defined as the value of any remuneration (salary, payment for services, consulting fees, honorarium, paid authorship, equity, option). The UMD threshold for reporting a FCOI is $0 as prescribed by Maryland State Ethics Law and University policy.
Institutional Conflict of Interest (ICOI) - An institutional conflict occurs whenever the financial interests of the institution, or of a University Official acting within their authority on behalf of the institution, might affect, or appear to affect, the objectivity of institutional processes for the selection, design, conduct, reporting, review, or oversight of the University enterprise. For the purposes of evaluating ICOI, activities related to research, teaching, and outreach, and the administration of those functions, singly and collectively, represent the University enterprise.
- University Officials covered under the University of Maryland's ICOI Policy are those officials who have the power to commit substantial resources of the University. University Officials include but are not limited to: the President, Assistant President, Vice Presidents, Associate and Assistant Vice Presidents, the Senior Vice President & Provost, Associate Provosts, Deans, Associate Deans, and Athletic Director, including those holding these positions in an interim capacity, as well as others who have discretionary authority to allocate resources related to the University enterprise as identified by any of the officials named previously. University Officials must disclose their significant economic interests and affiliations, and those of their immediate family members (spouse, domestic partner, or dependent) upon their appointment and then annually thereafter, and identify how those interests may relate to their institutional responsibilities.
Organizational Conflict of Interest (OCI) - As defined by the Federal Acquisition Regulations, an organizational conflict of interest may result when factors create an actual or potential conflict of interest on a Federal Government contract, or when the nature of the work to be performed on the contract creates an actual or potential conflict of interest on a future acquisition. In the latter case, some restrictions on future activities of the contractor may be required.
In other words, because of activities or relationships with other persons or organizations, an entity is unable or potentially unable to render impartial assistance or advice to the Government, the entity’s objectivity in performing the contract work is, or has the potential to be, impaired, and/or a person has an unfair competitive advantage.
What are the different types of Organizational Conflicts of Interest?
As outlined in the FAR Subpart 9.5, an OCI may exist in three basic categories:
- Unequal Access to Information: This type of OCI arises when the University gains an unfair competitive advantage because of a University Member’s access to information not generally available to other parties competing for the same federal funding. For example, a faculty member may have had access to budgets, statements of work, or evaluation criteria in the proposal submission and award process that were not available to other potential contractors.
- Impaired Objectivity: This type of OCI arises when a University Member’s work on a government contract places the University in a situation of evaluating itself or a related entity. For example, the University’s work under one government contract could require it to evaluate its own activities or evaluation of proposals from entities competing with the University. In this case, the concern is that the University cannot render impartial advice under a federal contract.
- Biased Ground Rules: This type of OCI arises when a University Member provides consultation, advice, or technical assistance relating to a federal funding opportunity, and the University then applies for that same funding opportunity. For example, a Faculty Member may serve on a federal advisory board or act as a consultant to develop standards, which are used as the basis for a Scope of Work or specifications that are used to support a federal funding opportunity.
What is the difference between Organizational Conflict of Interest (OCI) and personal Conflict of Interest (COI)?
Organizational Conflict of Interest looks at the institution and everyone in it, as well as any possible or actual conflicts. Personal Conflict of Interest, on the other hand, only looks at individuals. Removing someone from a project may solve a potential personal COI problem, but it will not solve a potential OCI problem.
Disclosing Presentations and Seminars inTERP
Instructions for how to download your Faculty Success report and uploading inTERP can be found here.
Conflict of Interest Management Plan Questions
Why must I update my inTERP disclosure when I already have a management plan in place?
UMD policy requires disclosures be updated annually and within 30 days of new or changed significant financial interests.
Why does my management plan require that I disclose my COI for publications and presentations?
It is important to disclose in publications and presentations to maintain a level of integrity and transparency in research reporting. Sample COI disclosure statement: "Dr. [UMD PI] co-founded XXXX to commercialize technology developed in collaborations with their labs. Company XXXX has an exclusive license from the University of Maryland for the commercialization of this technology."
Why does my management plan require that I disclose my COI to all working on my project?
Investigators share an obligation to identify and, when possible, avoid financial conflicts of interest. When conflicts cannot be avoided, investigators must disclose relationships that create, or reasonably appear to create, conflicts of interest, and work with university officials to manage or resolve those conflicts. All postdoctoral researchers, graduate students, and lab members, whether involved in the research/award or not, will be made aware of this COI in writing and will sign a memo indicating their acknowledgement. These signed acknowledgement documents will be filed with the UMD COI Office and kept up to date.
If I have a management plan, may UMD students work at my company?
No personnel employed by UMD will be employed part-time or full-time by the company, including undergraduate students, graduate students, post-doctoral researchers, or technicians. UMD employees will not conduct research for the company or be allowed in the company’s research facilities. UMD students employed by the company cannot be students in classes taught by UMD researcher(s) and/or be their advisees.
How should I disclose my outside interest in publications and presentations?
You should disclose your Conflict of Interest at the time of publication submission or within the initial slides of a presentation. Below are examples of language that can be used for such disclosures:
- Example 1 (equity interest): [Researcher’s name] has a financial interest in [Entity] and could benefit from the results of this research. This conflict has been reviewed by the University of Maryland and has been appropriately managed to maintain objectivity.
- Example 2 (licensed technology): [Researcher’s name] has a licensed [technology] to [Entity] and could benefit from the results of this research. This conflict has been reviewed by the University of Maryland and has been appropriately managed to maintain objectivity.
- Example 3 (consulting): [Researcher’s name] has a financial interest with [Entity] and could benefit from the results of this research. This conflict has been reviewed by the University of Maryland and has been appropriately managed to maintain objectivity.
How should I disclose my outside interest to my research team and collaborators?
A disclosure should be made at study initiation or as soon as a management plan is issued, whichever is first. As new members join the research team or collaborators join the project, a disclosure should be made to them. Below are examples of language that can be used for such disclosures:
- I have a financial interest in [Entity], and I could potentially benefit from the results of this research. To prevent any perceptions of bias or conflicts of interest, I am disclosing my financial interests to you and all of my research collaborators. This interest has been disclosed to and managed by the University's Conflict of Interest Office. If you have any questions or concerns, please feel free to contact me or the Conflict of Interest Office at any time.
- I have licensed [technology] to [Entity] and therefore I could potentially benefit from the results of this research. To prevent any perceptions of bias or conflicts of interest, I am disclosing my financial interests to you and all of my research collaborators. This interest has been disclosed to and managed by the University's Conflict of Interest Office. If you have any questions or concerns, please feel free to contact the Conflict of Interest Office at any time.
- I have a financial interest with [Entity], and I could potentially benefit from the results of this research. To prevent any perceptions of bias or conflicts of interest, I am disclosing my financial interests to you and all of my research collaborators. This interest has been disclosed to and managed by the University's Conflict of Interest Office. If you have any questions or concerns, please feel free to contact me or the Conflict of Interest Office at any time.
TO JOURNALS: (Name) (has financial and/or business interests in) (is a consultant to) (receives funding from) a company that may be affected by the research reported in the enclosed paper. S/He has disclosed those interests fully to the University of Maryland, and has in place an approved plan for managing any potential conflicts arising from (that involvement).
OR
This research is sponsored by (company A) and may lead to the development of products which may be licensed to (company B), in which (professor name) has a business and/or financial interest. S/He has disclosed those interests fully to the University of Maryland, and has in place an approved plan for managing any potential conflicts arising from this arrangement.
TO STUDENTS: For your information, I serve as (a consultant to) (a board member of) (an executive officer of) COMPANY NAME. I also receive research grant funding from (company name) investigating (briefly describe the research area). This relationship has been identified as having the potential to create a conflict of interest with my responsibilities as a faculty member. I have fully disclosed these interests to the University of Maryland, and I have in place an approved plan for managing any potential conflicts arising from this involvement.
I understand that your work on the project, (insert project name), should be for academic reasons to further your studies and your professional career endeavors. If at any time you have any concerns about whether your work is inappropriately focused toward my outside relationship(s), or that your ability to publish has been impeded in any way, I encourage you to contact the Department Chair or (alternative advocate for the student) (include names and phone numbers, if not evident).
I look forward to continuing to serve as your advisor and you can continue to count on my utmost support of your work.
What are some Familial Management Plan Best Practices?
Please review the Familial Management Plan Best Practices.
Conflict of Interest & Gifts Questions
Do I have to disclose gifts that support my research?
Yes, you must disclose all current or pending research gifts in support of your research program to you or through the University. Research gifts include funding and/or resources, for example, but not limited to: equipment, materials, software, and other items of value.
In the inTERP disclosure form, say yes to this question:

Check the anticipated or received gifts box.
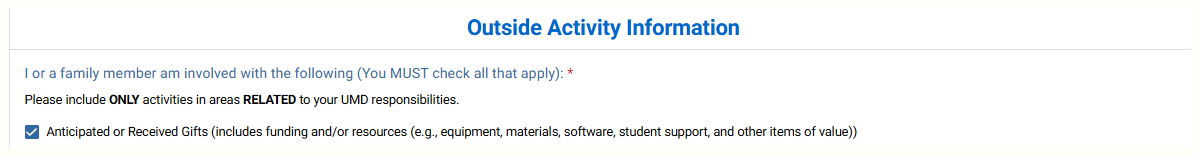
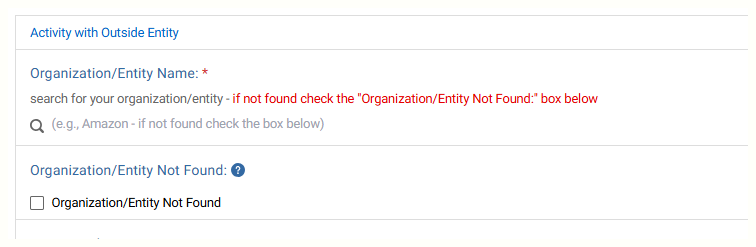
Enter the entity providing the gift and answer the questions.
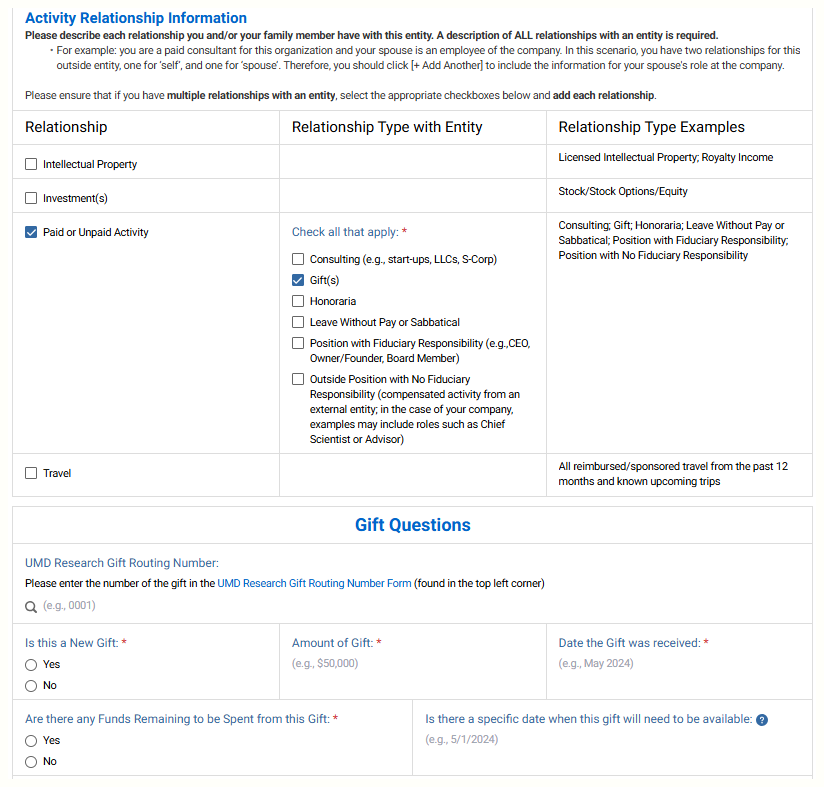
In the Activity Relationship Information, check paid/unpaid and gift. Answer the gift questions sections.
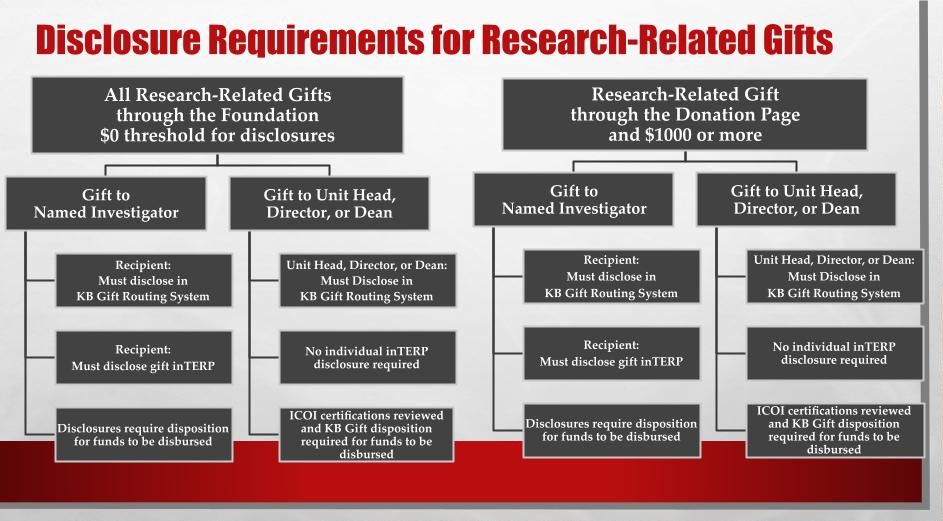
Where can I find more information about the required disclosures for Gifts?
 Questions regarding gifts should be directed to: giftvsgrant@umd.edu
Questions regarding gifts should be directed to: giftvsgrant@umd.edu
Conflict of Interest & Company Questions
I have an award from the Maryland Industrial Partnerships (MIPS) program. Is there something special for these projects?
All potential COIs on MIPS awards - disclosed at the time of application or not - must be resolved prior to award setup/expenditure.
Examples of potential COIs include (but are not limited to) situations in which the university PI, a family member, another UMD project researcher, or any other university employee:
- Holds equity ownership or equity option interest in the partner company (even if of no monetary value),
- Holds a company board membership (even if unpaid),
- Is an employee of, officer of, or consultant to the company (even if unpaid),
- Has any other contractual or financial relationship with the company.
Follow these instructions to include a MIPS proposal or award in your inTERP Disclosure.
Resolving Conflicts: Management Plans - Risk Level 2 for example plans for MIPS awards.
When I have a company and sponsored research, how do I account for my time?
When you list your company or any consulting entity, only enter the hours/week that you spend in your role supporting your company.
What if my company or an entity I am consulting with also has a joint award with my sponsored research agency?
Be sure to account for all hours spent in your consulting role ONLY when including the number of hours spent per week consulting. Hours spent supporting sponsored research in your role as a UMD researcher is NOT included in the total number of hours consulting.
How many 8 hours/week can I spend per company?
The TOTAL number of hours spent on ALL consulting activities outside of UMD is 8 hours per week. For example, you consult for three companies, two of which you own. You may not account for more than 8 hours total per week spent working for these companies.
I want to collaborate with a company. What do I need to consider?
Things to consider when collaborating with a company:
- Discuss the anticipated time commitment of the collaboration with Department Chair to avoid a Conflict of Commitment (see Conflict of Commitment Policy)
- You cannot be the PI on both the University and the company side. In a company sponsored collaboration in which you have a COI, the University employee may not represent the University in the research agreement.
- You may not employ students (with whom you have a University relationship, i.e. work in your group, are students in a course you currently teach, are your advisees, etc.) in the company
- Your company in the collaboration cannot use University facilities without an approved Facilities Use License (FUL)
- You must have an approved COI, FUL and Consulting MOU for UMD affiliate appointments of company personnel
- You cannot simultaneously accept a Sponsored Research Agreement or Gift through the UMD Foundation or another UMD Unit while serving as a consultant to the company providing the Gift
- If you are working as a consultant, what is the scope of work for the consultancy?
What you CANNOT do when you are both a UMD personnel and associated with a company
What you CANNOT do when you are both UMD personnel and associated with a company?
- Be PI on both sides of a proposal (no self-dealing).
- Solution: Pick a side for individual projects
- Have your advisees or lab personnel work in your company.
- Solution: Separate concurrent reporting lines
- Have students in your classes work in your company.
- Solution: Hire students only when they are no longer your students
- Serve on thesis/dissertation committees as a voting member if the student works at your company.
- Solution: Avoid concurrent arrangement
- Commingle lab and company activities.
- Solution: Keep space, people, and project tasks separate
- Give the University gifts, internships, etc to benefit your own research.
- Solution: Fund research through proper channels
- Negotiate with the University on behalf of the company
- Solution: Have outside counsel or another corporate officer handle
What you CAN do when you start a company
Your company CAN:
- License University IP
- Hire students
- Rent space on campus
- Have a prime-sub relationship with UMD/Company
BUT there are constraints in order to manage the relationship and mitigate COIs.
OUR BEST ADVICE: At the time of incorporation, you should disclose to the University (consulting policy).
Conflict of Interest & Facilities Use Applications
What is the relationship between COI and the Facilities Use Application?
Any company interested in utilizing space at the University of Maryland must complete a Facilities Use Application (FUA). There are sections on the FUA that concern Conflicts of Interest. There is a representative from the COI Office on the FUA Review Committee to ensure there are no conflicts that result from the use of University facilities. An inTERP disclosure must be dispositioned before an FUA can be approved.
Conflict of Interest & Leave Without Pay (LWOP) Questions
What must employees do in order to request Leave Without Pay?
All employees requesting a Leave Without Pay (LWOP) must disclose the LWOP in their inTERP disclosure. inTERP disclosures are reviewed at a portfolio level in order to make a determination regarding the employee’s status.
Conflict of Interest & Subrecipients Questions
How does a Subrecipient without an FCOI policy submit a disclosure under UMD's policy?
Please go to ORA’s site on FCOI Subcontractor Guidance and/or contact the Disclosure Office.
How do I get assistance with setting up a subawardee in our inTERP system (foreign or domestic)?
The subawardee will need to disclose in the inTERP system since they will be following the University of Maryland FCOI policy. Please keep the Disclosure Office updated by providing names of people who are subject to this mandate.
For Subrecpients governed by UMD’s FCOI policies and procedures, the following steps must be completed by the party indicated:
- STEP 1: [UMD PI & SUBRECIPIENT] All Investigators named by the Subrecipient on their FCOI certification must work with the UMD PI’s department to obtain UNPAID APPOINTMENT status in order to access inTERP through the Central Authentication Service (CAS).
- STEP 2: [SUBRECIPIENT] Subrecipient Investigators must individually complete a Disclosure in KCOI so that the UMD COI Committee can review for any Financial Conflicts of Interest (FCOI’s).
- STEP 3: [SUBRECIPIENT] Each of the Subrecipient Investigators must also successfully complete the online Conflict of Interest CITI Training Module provided by the University of Maryland and provide a certificate of completion to interp@umd.edu.
- STEP 4: [UMD COI OFFICE] Upon completion of all the above, the UMD-COI will confirm with the UMD Subaward Administrator that all Subrecipient investigators have been cleared by the Disclosure Office.
- STEP 5: [UMD SUBAWARD UNIT] Subaward is issued to the Subrecipient organization.
Conflict of Interest & Travel Questions
In terms of travel, what is required to be disclosed in the COI section of inTERP?
Reimbursed/Sponsored travel must be disclosed in COI section, except when reimbursement comes from UMD, U.S. Federal, state, or local governmental agencies, U.S. Institutes of higher education as defined at 20 U.S.C. 1001(a), U.S. Research institutes affiliated with Institutes of higher education, U.S. Academic teaching hospitals and medical centers
All travel sponsored or reimbursed by a foreign organization must be disclosed, regardless of the type of organization sponsoring the travel.
If I was provided work-based travel in my last job outside of UMD, do I need to include it in the PHS reporting?
Based on the regulatory guidance you should not need to include the travel if it was not "related to your institutional responsibilities". However, the regulations do not distinguish between "current" and "former" institutional responsibilities. That being said, the best course of action would be to include the travel with a short description. This way it is clearly noted and you are covered.
Conflict of Interest Training
How can I register for Conflict of Interest Training?
Register and enroll in the Conflict of Interest Mini-Course through CITI. Click on View Courses, select Add a Course, and scroll to Question 5: Conflict of Interest Mini-Course. This course and associated quizzes should take 30-60 minutes to complete. If you pass the quizzes, the training is good for four years. You will be sent an automatic reminder when your training is due to expire.
Conflict of Interest Examples & Scenarios
What are some examples of Conflicts?
Examples of activities and situations that present, or may appear to present, potential conflicts of interest include, but are not limited to:
- Situations in which financial, professional, or other personal considerations may directly or indirectly affect, or have the potential to affect, an employee’s professional judgment in exercising any university duty or responsibility in administration, management, instruction, research, or other professional activities. This can include interests that bias the nature or direction of scholarly research or influence decisions with respect to teaching and students, appointments and promotions, use of university resources, interactions with human subjects, or other matters of interest to the university.
- Situations in which outside relationships or activities interfere with, or have the potential to interfere with, an employee’s commitment to their university duties or responsibilities.
- An employee’s external personal entity/company utilizes, supports, or funds their sponsored research.
- An employee purchases via university or sponsored research funds any goods or services from his/her external personal entity/company.
- An employee’s external personal entity/company licenses intellectual property to, or otherwise profits from, sponsored research.
- An employee subcontracts any aspects of his/her sponsored research to his/her external personal entity/company.
- An employee’s Significant Interest in his/her external personal entity/company could directly and significantly affect the design, conduct, or reporting of their sponsored research or educational activities.
- Diversion of university business opportunities to an external entity/company.
- Assignment of students or graduate assistants to duties that benefit an external entity/company in which the person in charge of assignment of those duties has a Significant Interest.
- Use of university facilities, supplies, or personnel for the benefit of an external entity/company in which the employee has a Significant Interest.
- Assumption of responsibilities for an external organization that divert a university employee from his/her attention to university duties, or create other conflicts of loyalty.
- Use for personal profit of unpublished information emanating from university research or other confidential university sources, assisting an external organization by giving it exclusive access to such information, or consulting under arrangements that impose obligations that conflict with university patent policy or with the university’s obligations to research sponsors.
- Conduct of research that could and ordinarily would be carried on within the university elsewhere to the disadvantage of the university and its legitimate interests.
Can you provide some scenarios where a Conflict of Interest exists?
- A faculty member or Employee proposes to accept a Consulting position to assist an entity/company that seeks to compete directly with an UMD program.
- A faculty member or Investigator accepts a payment from an entity/company with which the faculty member or Investigator recommends a UMD contract.
- A faculty member or Investigator proposes that UMD enter into a business transaction with an entity/company in which the faculty member or Investigator (or his or her Immediate Family Member) has an Ownership Interest.
- A faculty member or Investigator proposes that UMD use or purchase a product, which would result in an economic benefit to the faculty member or Investigator or an immediate family member. For example, a faculty member advocates the use of software the faculty member created prior to joining UMD and as to which the faculty member receives a royalty based on the commercial use of the software.
- A faculty member or Investigator proposes that UMD enter into a technology licensing arrangement with an entity/company in which the faculty member or Investigator or an immediate family member serves on the board of directors or is employed.
- A faculty member or Investigator proposes that UMD hire or contract with a member of his or her family for a position that is within his or her direct or indirect supervision or control. UMD’s employment of a family member must be in accordance with UMD’s Policy on the Employment of Members of the Same Family.
- A faculty member or Investigator proposes to undertake or orient his or her research at UMD to serve the private interests of an outside entity/company without disclosure and permission.
- A faculty member or Investigator proposes to share unpublished data from UMD research with an entity/company as part of a consulting engagement or in discussions about a proposed consulting engagement.
Outside Professional Activity Examples
Outside Professional Activity Examples
Outside Professional Activities include any additional professional activities, that may be paid or unpaid, are beyond a University employee's institutional responsibilities, are within their discipline or area of expertise, and could include professional service
Committees, Panels, and Professional Organizations
- Chairing a master's or doctoral thesis committees at another university
- Engaging in other outside activities requiring a significant time commitment (e.g. boards, public office, etc.) or that have the potential to compete with my UMD responsibilities
- Leadership positions in professional societies
- Serving on an advisory committee or evaluation panel for government funding agencies, nonprofit foundations, or educational organizations
- Serving on but not chairing, master's or doctoral thesis committees at another university
- Service with accreditation agencies
Peer Reviews, Scholarly Publications, and Editorial Services
- Editorial services for educational or professional organizations
- Peer Review of articles and grant proposals
- Preparing scholarly publications
Outside Teaching, Presentations, and Seminars
- Conducting short courses including non-credit or professional courses
- Presenting or conducting a workshop at professional meetings or other similar gatherings
- Teaching academic or professional courses for another university/organization, including but not limited to online teaching
Scholarly Collaboration and Research
- Collaboration/research with an institution of higher education where there is no compensation, an agreement, a contract, or required time commitment
- Scholarly collaboration with an institution of higher education where there is no compensation, agreement, contract or required time commitment
Non-research related consulting
- Not related to your research