Animal Care and Use Express (CUE) Sheet
A bi-monthly (ish) communication with important information to keep you right on cue!
FY 25-26 | Issue 6
Semi-Annual IACUC Inspections Coming!
Spring semester semi-annual inspections begin in a few weeks (April 1)! Many of you are familiar with the process, but it is helpful to refresh your memory using information on our “Inspections” webpage or the accordion menu below.
Why do we perform inspections?
Federal regulations require us to inspect our facilities and review our program at least once every six months. In addition to meeting our regulatory requirements, inspections allow us to ensure the highest quality safety and health of the animals and personnel engaged in animal research activities.
These also provide us with an opportunity to identify programmatic and ongoing deficiencies before they become reportable non-compliance issues.
What spaces get inspected?
ALL areas in which live animals are housed, undergo procedures (such as surgery) or are euthanized and also…
ALL areas that are utilized for animal research support including storage areas, cage wash areas, corridors within dedicated large vivaria, imaging facilities, and more.
These areas include centralized and satellite facilities where animal care is provided by dedicated staff, as well as Principal Investigator, or PI-managed facilities where animal care is completely provided by the PI and/or other trained lab members.
Who performs the inspections?
Generally speaking, at least two members of the IACUC must be present at the inspection of each space, and all members of the IACUC are invited to participate in the inspection of any space.
For larger spaces, like the farms or our larger vivaria, the IACUC may use larger groups or sub-groups of inspectors who will look at different parts of the facility.
How can I prepare for an inspection?
Broadly, inspectors look for findings pertaining to information found in regulatory documents like the Animal Welfare Act and the PHS Policy, as well as publications like the Guide, the Ag Guide, and others. We also have institutional standards and guidelines.
UMD researchers and facilities personnel are expected to operate in compliance with these standards in an ongoing basis, so no special preparation should be required. We provide our Inspection Checklist publicly on our website and in IRBnet for all inspectors and researchers to refer to at any time, but it is especially important to familiarize yourself with the checklist before each inspection round.
We encourage you to perform a self-inspection of your spaces using our checklist, and we have provided some quick guidance from the Fall ’25 inspection data below the graphic.
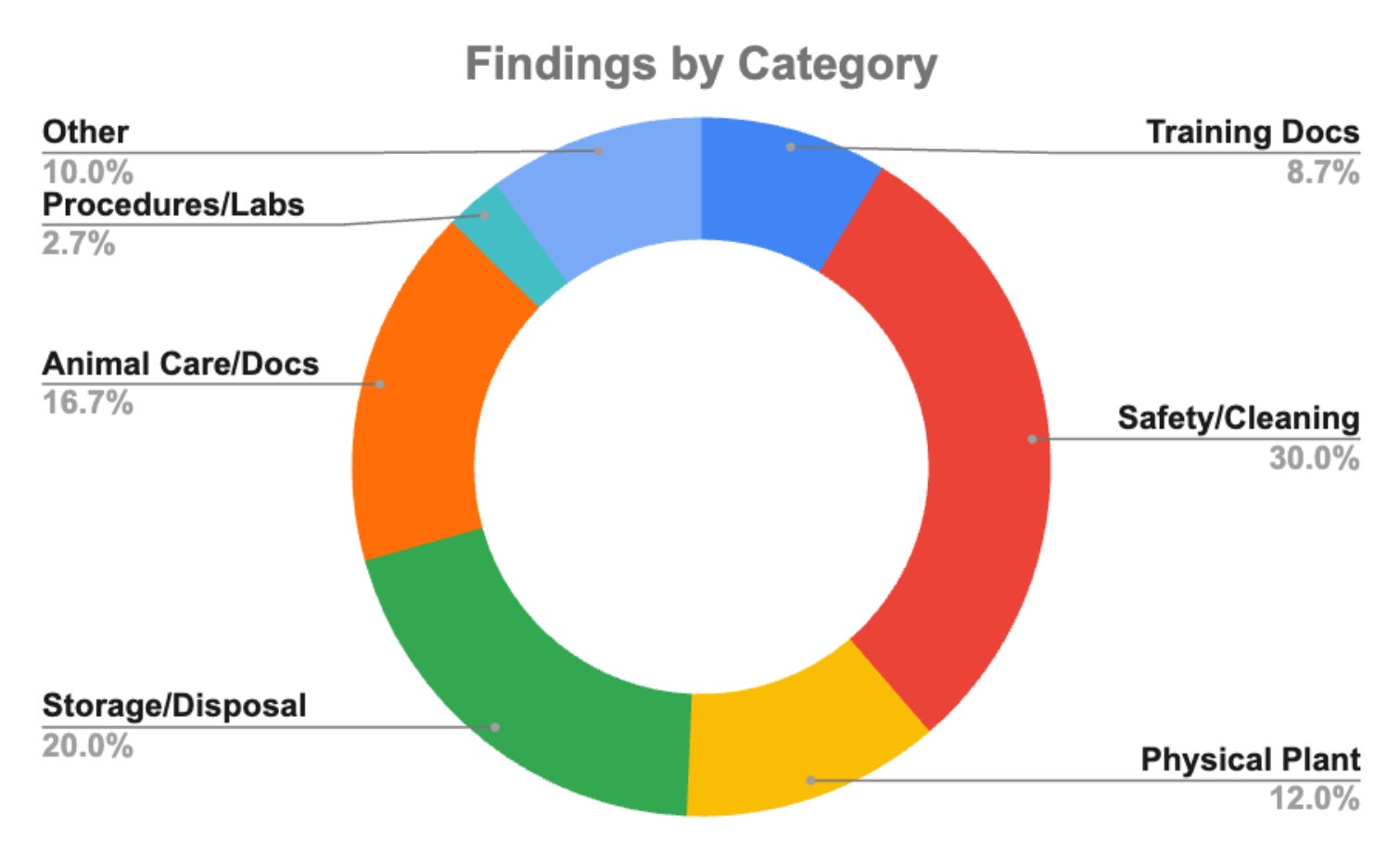
Noted for the Fall 2025 Inspection:
- Continued improvement in total number of findings (220 SP24 | 150 FA25)
- Significant improvement in training documentation findings. Automated emails from our database should help you stay on top of this admin task—please reach out if you have not received this to check the training status and enrollment for all of your current lab personnel.
- The training database does not replace your in-lab documentation, please keep up to date training documentation in a log in your lab. Ensure that new personnel are aware of lab procedures and have access to protocols and SOPs pertaining to your research.
- Improved in all categories except for Storage/Disposal—additional guidance on storage of controlled substances, mixtures and solutions, expendable supplies and medications, and shelf life of sterile agents forthcoming.
- Check expiration dates of drugs and other medical supplies, and ensure proper labeling if applicable (e.g., expired drugs labeled “not for use in survival surgeries”). Ensure that sanitation supplies are in date as well with labels for date opened/mixed as appropriate.
- Feed storage (and other husbandry supplies): Feed should be rotated first-in, first-out and clearly labeled with expiration/mill dates. Opened feed should be stored in sealable, sanitizable bins with clear expiration dates and sanitation dates. Temperature and humidity should be in appropriate ranges and recorded daily. Other clean husbandry items (water bottles, food/water bowls, toys, huts, etc.) should be covered and stored appropriately.
- Recordkeeping:
- Make sure sanitation records for lab spaces, animal housing areas and surgical suites are maintained appropriately. This includes euthanasia equipment maintenance and records.
- Cagewash and Autoclave: Make sure that test strips and other records are maintained and available. Ensure that emergency functions are working appropriately and that staff are trained on emergency procedures.
- Please check that previously cited physical plant deficiencies are repaired.
- Most Safety/Cleaning items related to general cleanliness, eyewash maintenance and documentation, and pest control.
- Ensure that your spaces are free of clutter, dust and debris, and that cleaning implements are not stored directly on the floor.
- Eyewash stations should be flushed weekly and documentation should be updated each time.
- Pest control traps should be maintained and documented. Issues should be reported to Travis Kramer at tkramer@umd.edu. Keep documentation of communications.
- Pest traps should be small, low-level adhesive insect traps that will not trap rodents.
- Animal health must be checked daily, and records must be updated and available.
- Sick and deceased animals must be reported to the AV’s office via appropriate channels.
Other Mid-Semester Reminders
- Website migration to a new content management system has been completed as of March 2, 2026. Any questions about new website content should be directed to iacuc@umd.edu. There are some content freezes anticipated, and we anticipate access issues as well.
- The AAALAC Site Visit is planned for the Fall 2026 semi-annual inspection timeframe and data collection for the Program Description will begin soon. Find more information here.
- A new PI-Specific Refresher Training course has officially rolled out. PIs who have already completed Initial PI/Animal User Training may register for the dates below.
Important Dates
| Meeting Dates: | Submission for Pre-Review by: |
|---|---|
| March 26, 2026 | Submission for pre-review has passed (February 15, 2026) |
| April 23, 2026 | March 15, 2026 |
| May 28, 2026 | April 15, 2026 |
| June 25, 2026 | May 15, 2026 |
| July 30, 2026 | June 15, 2026 |
NO AUGUST MEETING | The IACUC will try to send projects submitted during this time to DMR. Those called to FCR will go to September meeting.
Click buttons below lists to register!
New PI/Animal User Training is required for all new PIs, students, assistants, etc., who will work directly with animals. This includes all handling, husbandry, procedures, transportation, etc.
Click here to register for NEW PI/Animal User Training
NON-PI Refresher Animal User Training is required every 3 years in order to continue working with animals.
Click here to register for NON-PI Refresher Animal User Training
PI-SPECIFIC Refresher Animal User Training is required for all PIs with approved UMD IACUC protocols every 3 years in order to continue working with animals.
Click here to register for PI-SPECIFIC Refresher Animal Care Training
| New PI/Animal User Training | NON-PI Refresher Training | PI-SPECIFIC Refresher Training |
|---|---|---|
| March 19, 2026 at 1:30 pm | March 24, 2026 at 3:30 pm | March 25, 2026 at 2:30 pm |
| March 27, 2026 at 11:30 am | April 3, 2026 at 1 pm | April 7, 2026 at 8 am |
| March 31, 2026 at 1 pm | April 15, 2026 at 11 am | April 24, 2026 at 11:30 am |
| April 8, 2026 at 10 am | April 29, 2026 at 12 pm | May 7, 2026 at 2 pm |
| April 21, 2026 at 10 am | May 12, 2026 at 2 pm | May 18, 2026 at 7:30 am |
| April 30, 2026 at 11 am | May 27, 2026 at 10 am | June 2, 2026 at 3:30 pm |
| May 5, 2026 at 10 am | June 4, 2026 at 12 pm | June 17, 2026 at 9 am |
| May 15, 2026 at 10 am | June 15, 2026 at 11 am | June 29, 2026 at 2:30 pm |
| May 19, 2026 at 1:30 pm | July 2, 2026 at 9:30 am | |
| June 3, 2026 at 3 pm | ||
| June 9, 2026 at 11 am | ||
| June 16, 2026 at 11 am | ||
| June 23, 2026 at 12 pm | ||
| July 1, 2026 at 3 pm |
Dates and times are subject to change/cancellation.
